A court in Marseille, France, sentenced the founder
of a French company on Tuesday to four years in prison for selling hundreds of
thousands of defective breast implants in more than 65 countries.
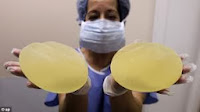
Jean-Claude Mas, 74, the founder of Poly Implant
Prothèse, and four of his former employees were found guilty last spring of
aggravated fraud after their company used a less expensive, industrial-grade
silicone to fill implants for a decade. The implants ruptured at a much higher
rate than the industry norm, leaking silicone into body tissues.
During the trial, which involved 7,400 civil
plaintiffs and 300 lawyers, Mr. Mas acknowledged that his company had used a
cheaper, unapproved product in its implants, but he argued that it was not
harmful.
More than 16,000 women have had their implants
removed since the scandal emerged in 2010. Poly Implant Prothèse, which was
founded in 1991, was closed by the French authorities in March 2010.
In addition to imposing the maximum prison sentence
on Mr. Mas, the court ordered him to pay a fine of 75,000 euros, or $103,000,
and sentenced his former employees to between 18 months and three years in
prison. Some of those sentences were suspended. Yves Haddad, Mr. Mas’s lawyer,
said that his client would appeal.
Last month a French court ordered a German
quality-control company to pay $4,000 each to 1,600 women who received
defective breast implants made by PIP. The German company, TUV Rheinland, was
accused of granting European Union safety certificates to the defective
implants.
About 300,000 women around the world received the
implants, which were not approved for sale in the United States.
A breast implant is a prosthesis
used to correct the size, form, and texture of a woman’s breast; in plastic
surgery, breast implants are applied for post–mastectomy breast reconstruction; for correcting congenital
defects and deformities of the chest wall; for aesthetic
breast augmentation; and for creating breasts
in the male-to-female transsexual
patient.
There are three general types of breast implant
devices, defined by their filler material: saline solution, silicone gel, and
composite filler. The saline implant has an elastomer silicone shell
filled with sterile saline solution; the silicone implant has an
elastomer silicone shell filled with viscous silicone gel;
and the alternative composition implants featured miscellaneous fillers, such
as soy oil, polypropylene string.



No comments:
Post a Comment